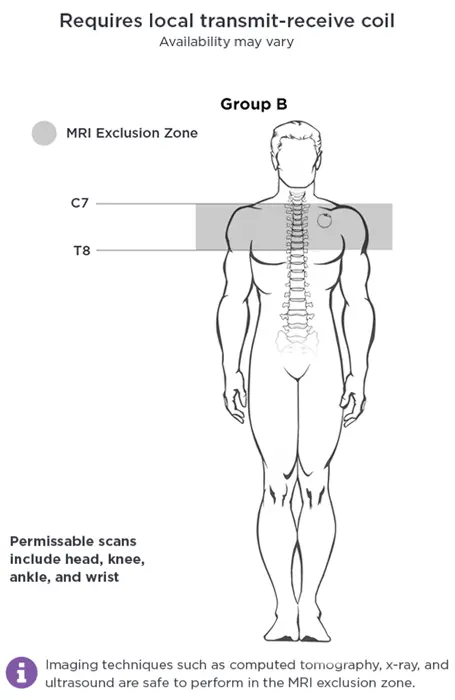
MRI Safety - Group B
Scan Conditions
Applies to all Pulse™ Model 102, Pulse Duo™ Model 102R, DemiPulse Duo™ Model 104
AND DemiPulse™ Model 103, AspireHC™ 105, ApsireSR™ Model 106, SenTiva™ Model 1000, SenTiva DUO™ Model 1000-D implanted lower than rib 4 (below armpit level)